But elements such as nitrogen are not very flammable and will not burn easily. For example oxygen will burn readily when heat is added. 
Some elements and compounds catch fire very easily and these will burn giving off heat energy. We can measure this by heating an object and seeing if it starts burning. However an element such as sodium is very reactive, when it is placed with other elements it will easily form compounds and react with them, sometimes releasing gases and heat energy.įlammability is how easy it is for an element to catch fire. For example elements like Neon, in the noble gases does not like reacting with other elements and does not want to form compounds. When we say chemicals are reactive this is a measure of how easy it is to get them to form compounds with other elements. 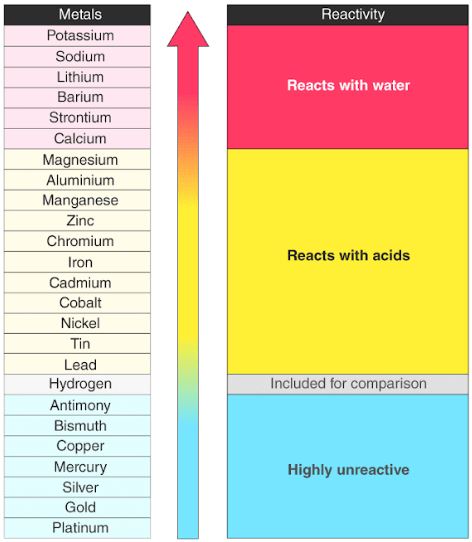
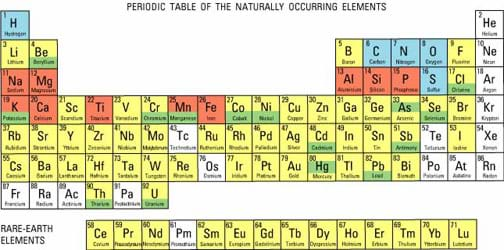
They do this by swapping or moving electrons, a particle in the atom which can be removed and added. ReactivityĮlements react together to form compounds. Where they are in the periodic table can be used to often predict the way they will react as they are in groups based on their similarities. When elements react they often bond together, make compounds, give out or take in heat or light and change from the elements they were into something new. It is attractive in colour and brightness, durable to the point of virtual indestructibility, highly malleable, and usually. Gold has several qualities that have made it exceptionally valuable throughout history. All elements have different ways they react together. gold (Au), chemical element, a dense lustrous yellow precious metal of Group 11 (Ib), Period 6, of the periodic table of the elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
